Messenger RNA species homologous to HepG2 glucose transporter messenger RNA were detected in K562 leukemic cells, HT29 colon adenocarcinoma cells, and human kidney tissue.
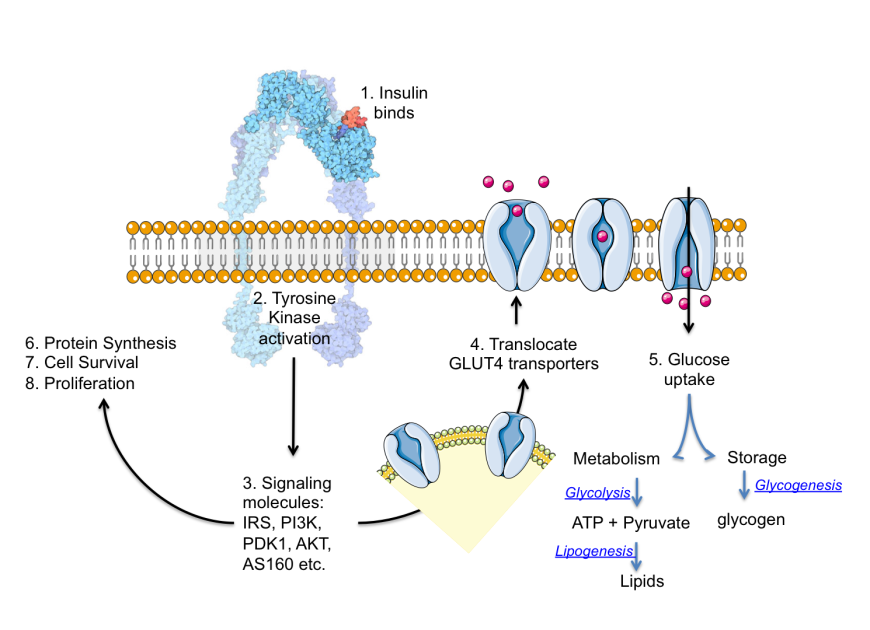
SGLT2 inhibitors are a class of prescription medicines that are FDA-approved for use with diet and exercise to lower blood sugar in adults with. The amino terminus, carboxyl terminus, and a highly hydrophilic domain in the center of the protein are all predicted to lie on the cytoplasmic face. Sodium-glucose Cotransporter-2 (SGLT2) Inhibitors. Most mammalian cells import glucose by a process of facilitative diffusion mediated by members of the Glut (SLC2A) family of membrane transport proteins. Several of these may form amphipathic α helices and contain abundant hydroxyl and amide side chains that could participate in glucose binding or line a transmembrane pore through which the sugar moves. bind to receptors produce their corresp Glucose induces insulin secretion. Glucose transporters (GLUT) function facilitated diffusion of glucose down concentration gradients with increasing glucose concentration, rate of transport assumes a hyperbolic curve due to carrier saturation divided into many subtypes that localize to different tissues GLUT-1 function basal glucose uptake. Glut1 has not been targeted until very recently due to the lack of potent and selective inhibitors. of Action Sodium-glucose co-transporter-2 inhibitors work by inhibiting. Analysis of the primary structure suggests the presence of 12 membrane-spanning domains. Glucose transporter (GLUT1) allele (XbaI) associated with nephropathy in non-insulin-dependent diabetes mellitus. The increased glucose transport in cancer cells has been attributed primarily to the upregulation of glucose transporter 1 (Glut1), 1 of the more than 10 glucose transporters that are responsible for basal glucose transport in almost all cell types. The protein lacks a cleavable amino-terminal signal sequence. Structural analysis of the purified human erythrocyte glucose transporter by fast atom bombardment mapping and gas phase Edman degradation confirmed the identity of the clone and demonstrated that the HepG2 and erythrocyte transporters are highly homologous and may be identical. The amino acid sequence of the glucose transport protein from human HepG2 hepatoma cells was deduced from analysis of a complementary DNA clone.
